大规模表达哺乳动物蛋白
2023.11.09
哺乳动物细胞作为蛋白的表达系统具有糖基化、乙酰化、硫酸化等翻译后修饰,其表达的重组蛋白在分子结构、理化性质和生物学功能方面最接近于天然的高等生物蛋白质分子,更有可能获得与天然分子相同的生物活性。因而产生的完整抗体和天然抗体一样,具有生物学活性,既可识别抗原又可激活补体系统。哺乳动物细胞易被重组的DNA转染,经过筛选可得到转化的细胞,具有遗传稳定性和可重复性.表达的产物分泌到培养基中易于纯化。利用哺乳动物细胞表达蛋白产物已广泛应用于生物制品工业,如病毒疫苗、抗体、干扰素、免疫调节剂、激素、和生长因子等的大量制备。根据目的蛋白表达的时空差异,可将哺乳动物细胞表达系统分为瞬时、稳定和诱导表达系统[3]。常用的细胞系有CHO细胞,BHK 细胞、Sp2/0,HEK293 等。
Mammalian cell expression has become the dominant recombinant protein production system for clinical applications because of its capacity for post-translational modification and human protein-like molecular structure assembly. While expression and production have been fully developed and Chinese hamster ovary cells are used for the majority of products both on the market and in clinical development, significant progresses in developing and engineering new cell lines, introducing novel genetic mechanisms in expression, gene silencing, and gene targeting, have been reported in the last several years. With the latest analytical methods development, more attention is being devoted towards product quality including glycol profiling, which leads to better understanding the impact of culture condition during production.
Important features of mammalian cells include their ability to perform post‐translational modifications and to secrete glycoproteins that are correctly folded and contain complex antennary oligosaccharides with terminal sialic acid. These covalent modifications may modulate the clinical efficacy of the protein (e.g., circulatory half‐life and biospecificity) or result in properties that are of interest for biochemical characterization—e.g., with respect to structural stabilization, functional groups, and biological role. Mammalian‐produced proteins are quality‐controlled through a process whereby the progress of incompletely folded, misassembled, and unassembled proteins into the secretory pathway is selectively inhibited (Hurtley and Helenius, 1989). The correctly processed material progresses and is generally secreted as fully active protein. Another important feature of mammalian cell expression has been the ability to provide proteins bearing sialyl glycoforms closely resembling glycoproteins produced by humans. In contrast, E. coli does not possess any glycosylation machinery and yeast tends to produce hypermannose‐type glycosylation. The yeast‐produced glycoproteins are rapidly cleared from the circulation by the liver and also may induce adverse immunogenic responses in humans.
CHO 细胞,全称 Chinese Hamster Ovary Cell,即中国仓鼠卵巢细胞系。1957 年,美国遗传学家 Theodore T. Puck 从波士顿癌症研究基金会的 George Yerganian 博士实验室得到一只雌性中国仓鼠,并分离出原始的 CHO 细胞。从此,CHO 在科学研究和医药工业领域逐步获得广泛的应用。如今它是用来表达外源蛋白较多也颇为成功的一类细胞。CHO 细胞属于成纤维细胞,既可以贴壁生长。也可以悬浮生长。
不同宿主细胞表达的重组蛋白其稳定性和蛋白糖基化类型不同,需根据要表达的目的蛋白选择最佳的宿主细胞。COS细胞是进行外源基因瞬时表达时用途最广的宿主,其重组载件易于组建,便于使用,而且对插入DNA的量或者采用基因组DNA序列的情况都没有什么限制,便于通过检测表达情况来确证cDNA的阳性克隆,也利于快速分析引入克隆化cDNA序列中的突变。CHO细胞则利于外源基目的稳定整合,易于大规模培养,能在无血清和蛋白的条件下生比,是用于真核生物基因表达软为成功的宿主细胞。已用于多种复杂的重组蛋白的生产。
CHO cells have been used extensively as a host for stable expression of proteins. A number of CHO mutants have been developed that provide the user with tools to examine the synthesis of DNA, RNA, and protein as well as protein secretion, protein glycosylation, and intermediary metabolism. The most popular CHO sublines (also see Urlaub and Chasin, 1980) are CHO DXB11 (dhfr /dhfr+ −) and CHO DG44 (dhfr −/dhfr −). The DXB11 cell was derived from CHO‐K1 in 1978 by Lawrence Chasin. The CHO‐K1 cells were originally derived in the 1950s, and the cells that were used in Chasin's laboratory for the development of the DXB11 line were obtained from Ted Puck and Fa‐ten Kao at the Eleanor Roosevelt Cancer Institute in Denver in 1970. The DG44 cell line contains a double mutation in the dhfr genes and is not capable of natural reversion to the dhfr phenotype. The DG44 cell line was derived from CHO pro3+− cells by Chasin in 1982. The CHO pro3− was derived from the cell line established in the 1950s and is sometimes referred to as CHO Toronto, because it was used extensively in that city by Louis Siminivitch and colleagues.
哺乳动物细胞表达外源重组蛋白可利用质粒转染和病毒载体的感染。利用质粒转染获得稳定的转染细胞需几周甚至几个月时间,而利用病毒表达系统则可快速感染细胞,在几天内使外源基因整合到病毒载体中,尤其适用于从大量表达产物中检测出目的蛋白。目前常用的可将目的基因导^哺乳动物细胞表达的病毒载体分为两类:整合型如SV40病毒载体、反转录病毒载体和游离型如痘苗病毒、腺病毒载体。利用Sindbis virus(SV)、Scmliki Forest virus(sFV)和痘苗病毒载体感染哺乳动物细胞表达的蛋白在结构与功能上与天然哺乳动物来源的蛋白更相似。Liljestrom等利用SFV病毒载体感染哺乳动物细胞获得的外源蛋白占细胞总蛋白的。载体的选择取决于外源基因的导人方式和其调控元件是否有利于转录和翻译。真核生物基因高表达载体必须具有如下调控元件:①原核DNA序列,包括能在大肠杆菌中自身复制的复制子,便于筛选含萤组细菌的抗生素抗性基躅,以及便于目的基因插入的限制性酶切位点。目前采州的哺乳动物细胞表达戟体大都带有来自pBR322的衍生质粒如pX[3、pBRd和pM[的原核序列;②启动子和增强子;③剪接信号;④终止信号和poIyA加尾信号。为了将含目的基因的载体导入哺乳动物功物细胞.还必须加入遗传选择标记。常用的标记基因有胸腺激酶(tk)基因、二氢叶酸还原酶(dh]r)基因、新霉素(neo)抗性基因、氯霉素乙酰基转移酶(cat)基因等dhfr还可作为共扩增基因使外源基因的表达产物增加。当培养基中逐新增加氨甲蝶呤(MTX)的浓度时,随着细胞对MTX抗体的增加。dhfr基因与外源基因均明显扩增。据文献报道,在不断提高的选择压力下,dhfr及侧翼序列能扩增至上千个拷贝,大大增加目的基因的表达水平。
Mammalian cells are the optimal system to produce recombinant human proteins as they contain a near native cellular environment. The low success rate (~9–27%) of acquiring soluble human proteins from bacterial expression hosts underscores the insufficiency of simpler protein expression systems (Ding et al., 2002). The human proteome is intrinsically far more complex than in prokaryotes. For instance, the human proteome is 40-fold larger, the average polypeptides length is twice that of bacteria, and multidomain proteins are 3-times more frequent (Rebeaud et al., 2021). Human proteins also have a higher occurrence of repeats, intrinsically disordered regions, and uniquely folded domains (Rebeaud et al., 2021). To correctly fold human proteins, mammalian expression hosts rely on sophisticated protein chaperones, diverse post-translational machineries, and comparatively slow translation rates (4–6 fold slower than bacteria) which allows additional time for polypeptides to fold co-translationally (Widmann and Christen, 2000). These unique features of mammalian expression hosts provide a superior environment in which to express recombinant mammalian proteins.
外源基因在哺乳动物细胞中的表达与多种因素有关,主要是启动子和增强子的强弱以及它们之间的搭配。启动子需包含两个识别序列:mRNA转录起始点和TATA盒。TATA盒位于转录起始值点上游25--30bp处,是引导RNA聚合酶在正确起始位点转录所必需的序列,即保证转录的精确起始。其他上游启动子元件常位于TATA盒上游100~200bp之间。其功能是调节转录的起始频率和提高转录效率。启动于和增强子受细胞类型的限制,在不同的细胞系中有很大不同,因此需根据宿主细胞的娄型选择不同的启动子和增强子以便于目的基因的高效表达。常用的启动子有SV40、Rou肉瘤病毒RSV和巨细胞病毒CMV的启动子。常用的增强子有Rous肉癯病毒基因长末端重复序列和人巨细胞病毒增强子。james等利用糖皮质类周醇诱导的鼠乳房瘤病毒(MMTV)启动子和鼠乳房痛病毒长末端重复序列(MMTV-LTR),在CHO细胞中表达分泌的碱性磷酸酶,产量为利用常规的SV40和CMV启动子的10倍,超过0.4mg/ml。另外,在哺乳动物细胞中已发现存在大量在低氧环境中可诱导转录的基因,如编码红细胞生成素(EPO)转铁蛋白、血红素加氧酶-1等的基因,它们都有一个共同的顺式作用元件(CGTG),有利于在5’或3’侧翼区的低氧诱导低氧诱导作用因子-1(HIF-1)和低氧反应增强子(HRE)结合,激活靶基因的转录,在低氧浓度下可使重组蛋白大量表达。据报道红细胞生成素启动子在1%氧浓度比在21%氧浓度下活陛增强100倍。这又是一种新的提高表达量的作用机制。
哺乳动物细胞对培养环境十分敏感,营养和生长因子缺乏、缺氧、病毒感染、毒性代谢物的积累、机械搅动以及培养压力的增加等很多固素都可诱导细胞凋亡。大量细胞的死亡严重影响蛋白的表达产量。现已证实,有些基因能抑制细胞凋亡,如Bcl-12和BcI-x能抑制许多因素诱发的细胞凋亡,以及某肿瘤细胞从贴壁培养转入悬浮培养而诱发的细胆凋亡。A1ison等报道,感染dSsVCAT病毒载体的BHKBcl-12和CHOBcI-x细胞生存期均延长数倍。BHKBel2、HHKBcI-x均使感染SFVIIl!的细胞恢复生长到对数生长期的细胞感染后可存活达9d,LL12的产量提高一倍。有些化学制剂也能抑制不同阶段的细胞凋亡,如乙酰半胱氨酸tNAC、吡咯烷二硫氨基甲酸酯(PDTC)等以及caspase抑制剂Z-VADfmk、YVAD,cmk等,从而大大增加表达蛋白的产量。如利用NAC和z-VADfmk的细胞培养,表达蛋白的产量分别提高2.2和3.9倍。细胞凋亡的抑制不仅延长细胞的存活期,提高蛋白的产量,而且有利于下游的纯化过程。
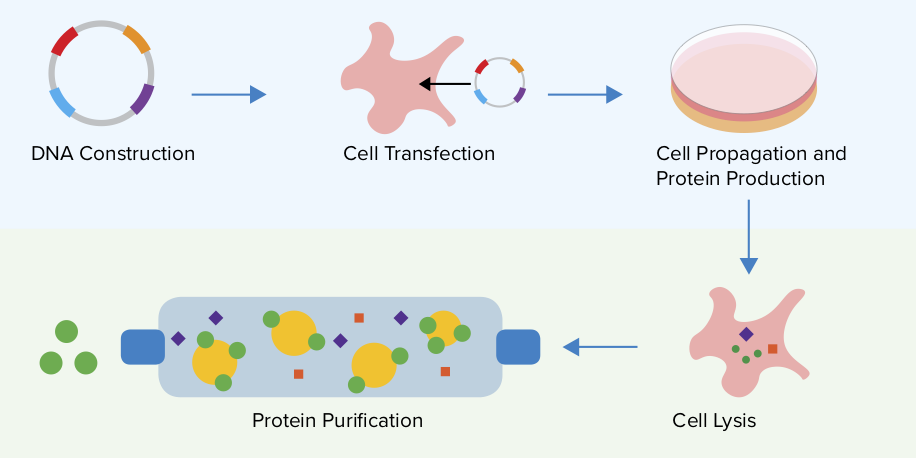
在细胞大规模培育过程,细胞的持续大量增殖使蛋白表达很快进入衰退期。同时由于营养缺乏,细胞大量死亡使细胞产品的产量下降,死亡的细胞释放蛋白酶和糖苷酶使表达产物降解,产品质量下降。为获得产物的大量有效表达,必须控制细胞在达到最适表达产物期停止增殖。现已发现四环素抑制表达系统(Tctoff)可严格控制细胞增殖过程。外源基因和抑制细胞生长的基因(如P27)在同一个双顺反子表逸单位,在可稠节的启动子(Totoff)作用下使细胞增殖期与增殖抑制产物表达期分开。细胞生长和产物表达都能达到最大程度。Totoff基因表达系统的优点在于毒性低,作用快,不影响哺乳动物细胞的代谢过程。由于四环素很不稳定,易于氧化脱水、芳香基化和表易构化作用,使Tot基因表达系统成为一个高效、无毒、具有严密开关功能(Trtswitch)的可诱导性基因表达系统,仅受四环素初浓度的影响,终产物中四环素自发降解,有利于下游的纯化过程。这种Tel基因表达系统控制的蛋白表达方式,因使细胞生长期和产物袁达期分开而适用于生产使细胞具有代谢负担或毒性的蛋白产物。mazur等报道利用该系统,在作用于细胞周期的激酶抑制子p27诱导下,使CHO细胞成功地达到持续生长抑制期,表达分泌型碱性磷酸酶SEAP(一种典型的外源糖蛋白)水平提高10~15倍。
在孵育批量培养中,通过减少葡萄糖和谷胱甘肽的量,使细胞代谢发生改变,以减少代谢产物乳酸、胺的形成,使细胞培养达到一种稳定状态,细胞浓度和蛋白产物大大增加。
目前的生物工程要求使用第三代无血清培养基,即无血清无蛋白(或含量极低)的双无培养基,使细胞培养很容易做到恒定,细胞分泌的的蛋白更易分离纯化,培养基的成本大为下降。随着基工程技术研究的深人,利用哺乳动物细胞表达蛋白产物的有效方式必将得到进一步发展,其在工业生产中的开发应用必将促进基因工程药物的大量生产。
我公司搭建的哺乳动物细胞蛋白表达平台,通过载体构建,筛选高产稳转细胞株,单批次可表达200L,成本优势明显。


